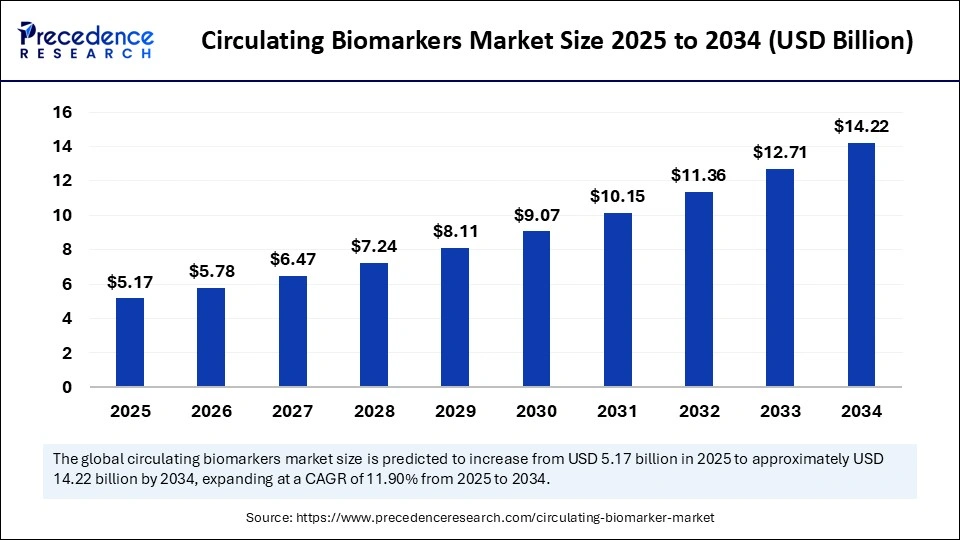
The global circulating biomarkers market size is projected to grow from USD 5.17 billion in 2025 to an impressive USD 14.22 billion by 2034, expanding at a robust CAGR of 11.90%. This growth is fueled by rising demand for early and non-invasive disease detection, increasing prevalence of chronic diseases, and breakthroughs in biomarker technologies.
Circulating Biomarkers Market Key Insights
-
Market valued at USD 5.17 billion in 2025 with a forecast to reach USD 14.22 billion by 2034, growing at 11.90% CAGR.
-
North America leads the market in size, while the Asia Pacific is the fastest-growing region.
-
Circulating DNA dominates by type, with circulating tumor cells expected to record notable growth.
-
Hospitals are the primary users of circulating biomarkers, though medical research centers are rapidly expanding.
-
Major companies include Abbott Laboratories, Becton Dickinson, GE Healthcare, Epigenomics AG, Agilent Technologies, and Biocept.
What Are Circulating Biomarkers, and Why Are They Important?
Circulating biomarkers are biological molecules found in bodily fluids like blood, urine, and saliva, providing critical information about health and disease status. These include circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), cell-free RNA, extracellular vesicles, and proteins. Their minimally invasive nature allows for early disease diagnosis, continuous monitoring, and personalized treatment strategies, especially vital for managing cancer and other chronic conditions.
How Is Artificial Intelligence Transforming the Circulating Biomarkers Market?
Artificial intelligence (AI) is revolutionizing this market by enhancing the analysis of complex biomarker data. AI algorithms efficiently process high-throughput sequencing and mass spectrometry data to identify novel biomarkers, improving diagnostic accuracy and prognostic predictions.
AI-powered liquid biopsies enable earlier detection of minimal residual disease, making personalized treatment adaptations more timely and effective. For instance, PanGIA Biotech’s AI-integrated urine-based liquid biopsy for prostate cancer is a notable breakthrough, commercialized in India in late 2024.
What Are the Key Drivers Behind Circulating Biomarkers Market Growth?
The circulating biomarkers market is expanding due to several factors:
-
Increasing prevalence of cancer and chronic diseases requiring precise monitoring.
-
Growing demand for precision and personalized medicine approaches.
-
Surge in regulatory approvals of novel biomarker tests enhancing clinical adoption.
-
Rising investments in healthcare infrastructure, research, and innovative technologies.
-
Development of advanced liquid biopsy and high-throughput analytical technologies.
Circulating Biomarkers Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 5.17 Billion |
| Market Size in 2026 | USD 5.78 Billion |
| Market Size by 2034 | USD 14.22 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 11.90% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Type, Application, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
What Opportunities and Trends Are Emerging in the Circulating Biomarkers Market?
Which regions offer the highest growth potential?
Asia Pacific is anticipated to grow fastest, driven by developing healthcare infrastructure, increasing R&D investments, growing cancer incidence, and expanding adoption of non-invasive diagnostic methods.
How are hospital and research sectors differing in their use of circulating biomarkers?
Hospitals dominate the current market by utilizing biomarkers for early disease detection and monitoring therapies. Meanwhile, medical research centers are accelerating growth by focusing on biomarker discovery and translating findings into commercial tests and treatments.
What breakthroughs are shaping the future of circulating biomarkers?
FDA approvals of innovative tests such as Guardant Health’s Shield blood test for colorectal cancer and Mercy BioAnalytics’ extracellular vesicle-based ovarian cancer test are driving clinical adoption. Strategic partnerships and AI integration are also accelerating innovation and accessibility.
Circulating Biomarkers Market Regional and Segmental Analysis
North America remains the largest market owing to heavy healthcare spending, high cancer prevalence, and robust R&D investments. The U.S. leads this dominance with advanced healthcare infrastructure and regulatory backing. Asia Pacific’s rapid growth is propelled by emerging markets focused on healthcare expansion and technology adoption.
By type, circulating DNA leads due to its established role in oncology for molecular tumor profiling. Circulating tumor cells are poised for rapid growth as chip and detection technologies advance.
Application-wise, hospitals currently dominate, especially through liquid biopsy diagnostics for cancer. Medical research centers, however, are emerging as key players in biomarker development for broader disease applications.
Latest Breakthroughs and Key Players
-
Guardant Health’s FDA-approved blood test for colorectal cancer screening (2024)
-
Mercy BioAnalytics’ breakthrough device designation for ovarian cancer screening (2024)
-
Myriad Genetics’ molecular residual disease test showing promise in renal cell carcinoma (2025)
-
Lumipulse’s FDA-cleared diagnostic for early Alzheimer’s detection (2025)
Top Key Players in the Circulating Biomarkers Market & Their Offerings:
- Abbott Laboratories: Abbott Laboratories is a global leader in diagnostics and molecular testing, offering advanced platforms for oncology, infectious disease, and genetic screening. The company’s innovations in liquid biopsy and molecular diagnostics provide precise, minimally invasive testing solutions. Abbott’s continuous investment in automation and data integration strengthens its position in personalized medicine and early disease detection.
- Becton, Dickinson and Company: Becton, Dickinson and Company (BD) specializes in molecular diagnostics and laboratory automation, delivering reliable systems for sample preparation, amplification, and analysis. Its BD MAX and BD COR platforms are widely used in clinical laboratories for high-throughput testing. BD’s focus on diagnostic accuracy and workflow efficiency supports its leadership in healthcare innovation.
- GE Healthcare: GE Healthcare integrates advanced imaging and molecular diagnostics technologies to improve disease detection and monitoring. Its AI-enhanced platforms support precision diagnostics and personalized treatment planning. GE continues to expand its molecular diagnostics capabilities through partnerships and R&D in oncology and genomic testing.
- Epigenomics AG: Epigenomics AG is a molecular diagnostics company focused on developing blood-based epigenetic tests for cancer detection. Its Epi proColon test is one of the first FDA-approved liquid biopsy assays for colorectal cancer screening. The company leverages DNA methylation biomarkers to create non-invasive diagnostics that enable early disease detection.
- Agilent Technologies: Agilent Technologies provides analytical instruments and molecular diagnostic solutions used in genomics, proteomics, and clinical research. Its technologies support biomarker discovery, precision oncology, and genetic testing. Agilent’s focus on high-sensitivity molecular analysis makes it a key enabler of advanced diagnostic research.
- Biocept: Biocept specializes in liquid biopsy diagnostics for cancer detection and monitoring. Its Target Selector platform enables the capture and analysis of circulating tumor cells (CTCs) and cell-free DNA from blood samples, offering insights into tumor evolution and treatment response.
- Affymetrix: Affymetrix, now part of Thermo Fisher Scientific, is known for its microarray and genomic profiling technologies used in biomarker discovery and molecular diagnostics. Its tools play a critical role in understanding genetic variation and disease pathways.
- Fluxion Biosciences: Fluxion Biosciences develops cutting-edge microfluidic systems for liquid biopsy, cell analysis, and single-cell genomics. Its IsoFlux and BioFlux systems enable high-sensitivity detection of rare cells and molecular biomarkers, supporting cancer research and precision diagnostics.
Challenges and Cost Pressures
Key challenges include high development and operational costs of advanced biomarker assays, reimbursement uncertainties, and the need for standardized protocols. Additionally, integrating complex biomarker data into clinical workflows poses technical and logistical hurdles.
Case Study Highlight
OncoHost’s EU-funded development of NeutroFlow, a biomarker predicting cancer patient responses to immunotherapy, exemplifies collaborative innovation. Leveraging consortium partnerships, they are advancing personalized cancer treatment and demonstrating the power of targeted biomarker applications.
Segments Covered in the Report
By Types
- Circulating DNA
- Circulating Tumor Cells
- Others
By Application
- Hospital
- Medical Research Center
- Others
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Read Also: Type 2 Diabetes Management Market
You can place an order or ask any questions. Please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344
- Managed DNS Services Market Size to Hit USD 2,288.60 Million by 2035 - March 5, 2026
- Power EPC Market Size to Hit USD 1,376.29 Billion by 2035 - March 5, 2026
- Ursolic Acid Market Size to Hit USD 68.77 Billion by 2035 - March 5, 2026
