What is the Pharmaceutical Stability And Storage Services Market Size in 2026?
The global pharmaceutical stability and storage services market is witnessing steady expansion, with its valuation expected to grow from USD 1.92 billion in 2024 to approximately USD 2.80 billion by 2030, registering a CAGR of 6.48% between 2025 and 2030. The growth trajectory is being fueled by the increasing complexity of drug formulations, a surge in biologics and biosimilars, and the rising need for regulatory-compliant storage and testing environments.
Pharmaceutical companies are increasingly outsourcing stability testing and storage operations to specialized service providers to ensure product integrity, reduce operational costs, and meet global compliance standards. The rapid expansion of clinical pipelines and heightened emphasis on patient safety are further strengthening demand.
What’s Driving the Market?
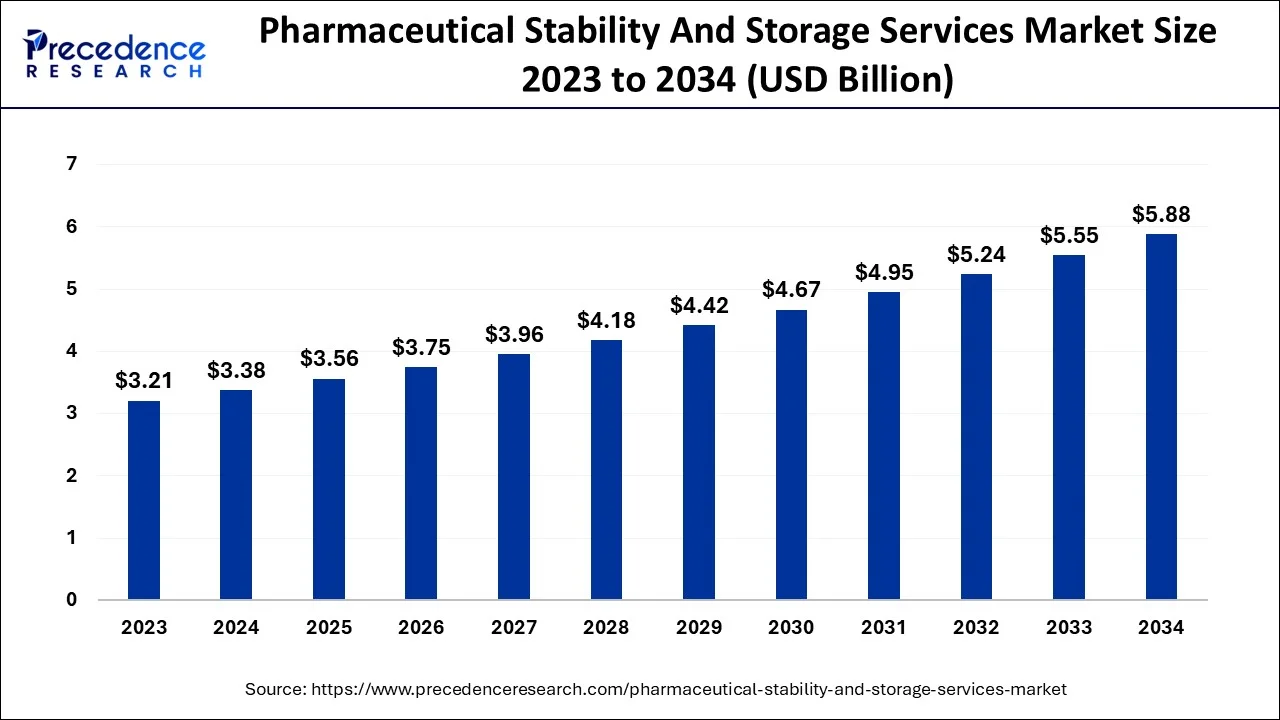
The market was valued at USD 1.92 billion in 2024 and is projected to reach USD 2.80 billion by 2030, reflecting consistent industry demand.
North America dominates the market, accounting for around 40% share, due to advanced infrastructure and strong pharmaceutical presence.
Asia Pacific is the fastest-growing region, supported by expanding pharma manufacturing and outsourcing trends.
Stability testing services lead the market, contributing over 73% of total revenue.
Small molecule drugs dominate, accounting for the majority of testing demand due to widespread therapeutic use.
How is AI Transforming Pharmaceutical Stability and Storage Services?
Artificial Intelligence is playing a transformative role in enhancing pharmaceutical stability and storage services. Advanced AI algorithms are now being used to predict drug degradation patterns, optimize storage conditions, and analyze large volumes of stability data in real time. This reduces the need for prolonged testing cycles and accelerates time-to-market for new drugs.
Moreover, AI-driven monitoring systems integrated with IoT-enabled storage units allow real-time tracking of temperature, humidity, and environmental variables. These smart systems not only ensure compliance with global regulatory frameworks but also minimize product loss and improve operational efficiency across storage facilities.
What are the Key Growth Drivers Shaping the Market?
1. Expanding Global Drug Pipeline
The increasing number of drugs under development—exceeding 9,000 globally—is significantly boosting the demand for stability testing and specialized storage services.
2. Rise of Biologics and Biosimilars
Biologics require stringent storage conditions such as ultra-low temperatures and controlled environments, thereby increasing reliance on specialized service providers.
3. Regulatory Complexity Across Regions
Diverse regulatory requirements across markets are encouraging pharmaceutical companies to outsource stability testing to ensure compliance and reduce delays in approvals.
4. Focus on Patient Safety and Product Integrity
Maintaining drug potency and safety throughout shelf life has become a critical priority, driving adoption of advanced stability testing solutions.
What Opportunities and Trends are Emerging in the Market?
Is Outsourcing Becoming the New Norm?
Yes. Pharmaceutical companies are increasingly outsourcing stability testing and storage services to reduce infrastructure costs and access specialized expertise.
Are Advanced Storage Technologies Reshaping the Industry?
Absolutely. Innovations such as real-time monitoring systems, automated stability chambers, and temperature-controlled logistics are transforming service capabilities.
Is Asia Pacific the Next Growth Hub?
Indeed. Rapid expansion of pharmaceutical manufacturing in countries like India and China is making Asia Pacific a lucrative market for service providers.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 3.56 Billion |
| Market Size in 2026 | USD 3.75 Billion |
| Market Size by 2035 | USD 6.1 Billion |
| Growth Rate from 2026 to 2035 | CAGR of 5.53% |
| Largest Market | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Services, Molecule, Mode and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America and Middle East & Africa |
Market Dynamics
Drivers
Rising demand for vaccines
The demand for vaccines—especially those that remain stable over longer periods—is increasing as governments continue to expand immunization programs. This trend became particularly evident during the COVID-19 pandemic and is expected to persist as countries strengthen their healthcare preparedness.
At the same time, pharmaceutical companies are significantly increasing their investment in research and development (R&D). This rise in spending is leading to a larger pipeline of drugs, each of which must undergo rigorous stability testing before progressing through clinical trial phases. As a result, the need for stability and storage services is growing steadily.
Growing demand for biosimilars
Biosimilars are gaining traction due to their similarity to biologic drugs and their comparatively lower cost. They are widely used in treating cancer, autoimmune disorders, and other chronic conditions. With the global burden of such diseases increasing, the demand for biosimilars—and consequently their storage and stability testing—is expected to rise.
Additionally, marketing pharmaceutical products across different regions is complex due to varying regulatory requirements. This complexity is pushing companies to rely more on outsourcing partners for stability testing and compliance support.
Restraints
Shortage of highly skilled professionals
One of the key challenges in this market is the limited availability of skilled professionals. Stability testing requires specialized expertise, and industries such as pharmaceuticals, biotechnology, contract research, and academia are all competing for the same talent pool.
Moreover, the shift toward more complex drug formulations and innovative molecular structures has intensified the demand for expertise. To address this gap, stronger collaboration between pharmaceutical companies, CROs, and research institutions is essential, particularly in developing targeted training programs.
Opportunities
A major emerging trend is the development of standardized global stability testing protocols. Companies are working toward creating unified testing conditions that can satisfy multiple regulatory requirements simultaneously.
For example, some firms are extending accelerated testing periods from 6 to 12 months and introducing more stringent environmental conditions, such as higher temperature and humidity levels. This approach allows companies to:
Avoid duplicate testing across regions
Optimize resource utilization
Speed up time-to-market
Additionally, combining environmental factors like temperature, humidity, and light in testing has proven to better simulate real-world conditions, improving the reliability of stability data.
Impact of COVID-19
The COVID-19 pandemic significantly increased the demand for vaccine storage and stability services worldwide. Governments accelerated vaccination campaigns, driving the need for robust cold-chain infrastructure and long-term storage solutions.
At the same time, pharmaceutical R&D activity surged, leading to more drugs entering clinical trials. Since stability testing is required at every stage of development, this has further boosted market growth.
The pandemic also contributed to a rise in certain health conditions. For instance, studies have shown a notable increase in pediatric diabetes cases between 2018 and 2021. This rise in disease prevalence is expected to increase drug production, indirectly driving demand for stability and storage services.
Service Insights
Stability testing dominated the market in 2023, accounting for over 63% of total revenue. This is because it is a mandatory requirement for regulatory approval at every stage of drug development.
Meanwhile, the storage segment is expected to grow at the fastest rate. Pharmaceuticals must be stored under controlled conditions—both cold and non-cold—to ensure their effectiveness over time. The growing number of small-molecule drugs, many of which do not require cold storage, is further driving demand in this segment.
Molecule Insights
Small molecules held the largest market share in 2023, contributing over 55%. These drugs are widely used to treat common conditions such as fever, migraines, cancer, and diabetes, making them a dominant segment in pharmaceutical development.
On the other hand, large molecules (biologics) are expected to grow at the fastest rate. Their increasing use in treating complex conditions like cancer, autoimmune diseases, and infectious diseases is fueling demand for specialized stability and storage solutions.
Mode Insights
In-house services accounted for the largest share in 2023, as many pharmaceutical companies prefer to manage stability testing internally for cost efficiency and control over processes.
However, outsourcing is expected to grow more rapidly in the coming years. The complexity of regulatory requirements across regions—despite alignment under global guidelines—makes outsourcing an attractive option for ensuring compliance and efficiency.
Regional Insights
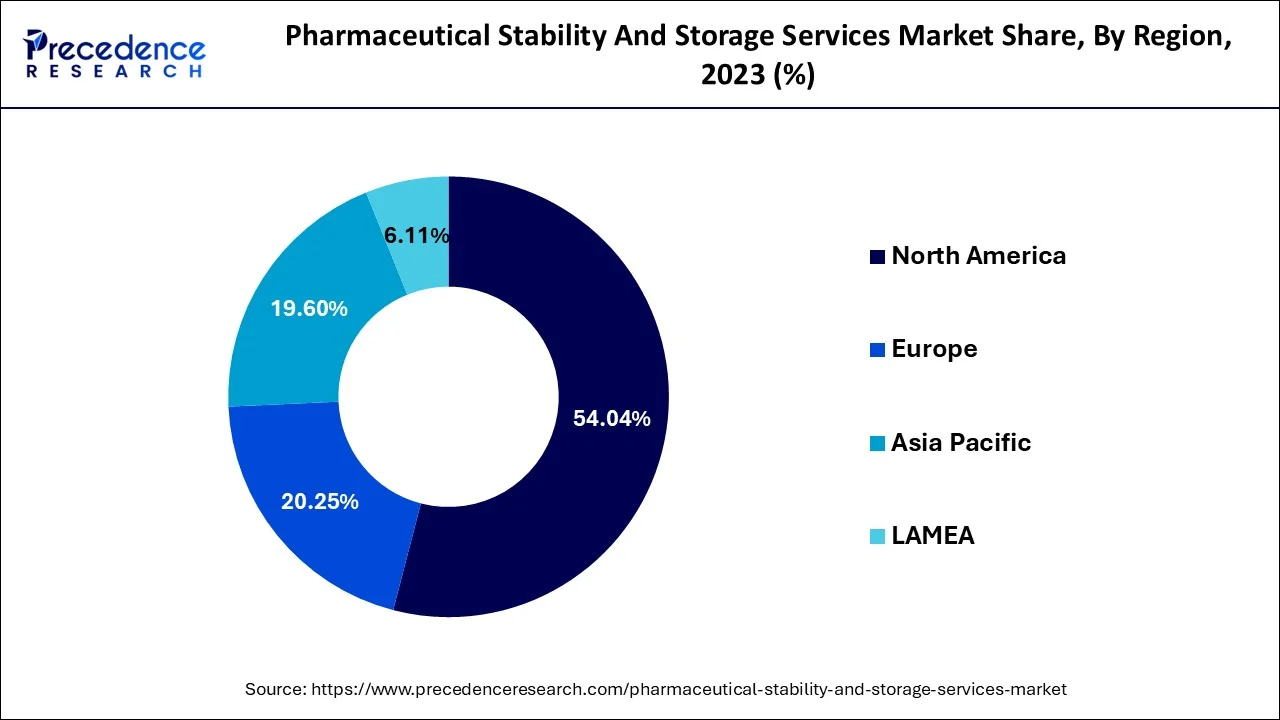
North America (Leading Market)
North America dominates the market, driven by a strong pharmaceutical industry and advanced infrastructure. The United States, in particular, leads due to strict regulatory frameworks and significant R&D investments.
Asia Pacific (Fastest Growth)
The Asia Pacific region is expected to grow at the fastest rate, supported by expanding pharmaceutical markets in countries like China, India, and Japan. Increased clinical trial activity and government initiatives to improve drug accessibility are key growth drivers.
India stands out due to its strong generic drug industry, regulatory compliance, and growing focus on biosimilars. Investments in cold-chain logistics have also strengthened its position globally.
Europe
Europe’s growth is driven by stringent regulatory standards and a mature pharmaceutical ecosystem. Increasing demand for biologics and sustainability-focused infrastructure improvements are key factors.
Germany plays a central role due to its strong manufacturing base, R&D capabilities, and strategic location for regional distribution.
Latin America
Latin America is emerging as a growing market, supported by improved regulatory frameworks and increasing participation in global clinical trials. Investments in cold-chain infrastructure are gradually enhancing service reliability.
Brazil leads the region with a well-developed pharmaceutical sector and growing demand for biosimilars.
Middle East & Africa
This region is experiencing steady growth due to healthcare modernization and rising pharmaceutical imports. Demand for cold-chain logistics and stability testing is increasing alongside vaccine distribution programs.
Saudi Arabia is a key contributor, driven by strong healthcare investments and efforts to localize pharmaceutical manufacturing.
Pharmaceutical Stability and Storage Services Market Companies
- Catalent, Inc.
- Charles River Laboratories International, Inc
- Almac Group
- Eurofins Scientific
- Lucideon Limited
- Intertek Group Plc
- Alcami Corporation
- Element Materials Technology
- Q1 Scientific
- BioLife Solutions
- Masy BioServices
- Roylance Stability Storage Limited
- Reading Scientific Services Ltd.
- Als Ltd.
- Auriga Research Private Limited
- Q Laboratories
- Precision Stability Storage
- PD Partners
Market Segmentation
By Services
- Stability
- Drug Substance
- Stability indicating method validation.
- Accelerated stability testing.
- Photostability Testing
- Other stability testing methods
- Storage
- Cold
- Non-cold
By Molecule
- Small Molecule
- Research Products
- Commercial Products
- Large Molecule
- Research Products
- Commercial Products
By Mode
- In-house
- Outsourcing
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- The Middle East and Africa
Get Sample Link: https://www.precedenceresearch.com/sample/2632
Also Read : https://www.reportsgazette.com/medical-device-outsourcing-market-2/
- Corporate Wellness Market Surges Toward USD 500.09 Billion by 2035 - April 22, 2026
- Portable Medical Devices Market Surges Toward USD 221.71 Billion by 2035 - April 22, 2026
- Infant Incubator Market Set to Surpass USD 3.27 Billion by 2035 - April 22, 2026
