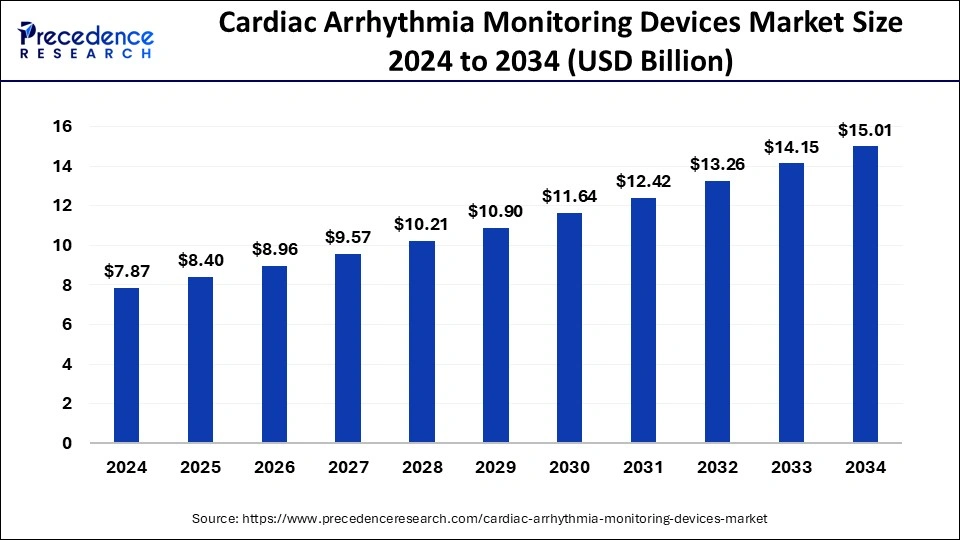
The global cardiac arrhythmia monitoring devices market size is estimated to rake around USD 14.15 billion by 2033 from USD 7.37 billion in 2023, growing at a CAGR of 6.74% from 2024 to 2033.
The cardiac arrhythmia monitoring devices market is witnessing significant growth globally, driven by the increasing prevalence of cardiovascular diseases (CVDs) and the rising geriatric population. Cardiac arrhythmias refer to irregular heartbeats that can lead to serious complications such as stroke, heart failure, or sudden cardiac arrest. As awareness about the importance of early detection and monitoring of arrhythmias grows, the demand for cardiac arrhythmia monitoring devices continues to rise. These devices play a crucial role in diagnosing and managing various types of arrhythmias, thereby improving patient outcomes and reducing healthcare costs associated with complications.
Key Points
- North America dominated the market with the largest market share of 37% in 2023.
- By device, the holter monitor segment has held the major market share of 43% in 2023.
- By application, the tachycardia segment dominated the market largest market share in 2023.
- By end-use, the hospitals and clinics segment has accounted more than 46% market share in 2023.
Growth Factors
Several factors contribute to the growth of the cardiac arrhythmia monitoring devices market. One key factor is the increasing incidence of cardiovascular diseases, including arrhythmias, attributed to sedentary lifestyles, unhealthy dietary habits, and a rise in risk factors such as obesity and diabetes. Moreover, advancements in technology have led to the development of innovative monitoring devices with enhanced accuracy, portability, and connectivity features, driving market growth. Additionally, growing investments in healthcare infrastructure and rising healthcare expenditure in both developed and developing countries are fueling the adoption of cardiac arrhythmia monitoring devices.
Get a Sample: https://www.precedenceresearch.com/sample/3948
Region Insights:
The cardiac arrhythmia monitoring devices market exhibits regional variations influenced by factors such as healthcare infrastructure, economic development, and prevalence of cardiovascular diseases. North America dominates the market, owing to a high prevalence of arrhythmias, well-established healthcare systems, and favorable reimbursement policies. Europe follows closely, with increasing awareness about cardiac health and a growing elderly population driving market growth. In the Asia-Pacific region, rapid urbanization, changing lifestyles, and improving access to healthcare services are contributing to the expansion of the market. Emerging economies in Latin America and the Middle East are also witnessing steady growth due to improving healthcare infrastructure and rising disposable incomes.
Cardiac Arrhythmia Monitoring Devices Market Scope
| Report Coverage | Details |
| Growth Rate from 2024 to 2033 | CAGR of 6.74% |
| Global Market Size in 2023 | USD 7.37 Billion |
| Global Market Size by 2033 | USD 14.15 Billion |
| U.S. Market Size in 2023 | USD 2.05 Billion |
| U.S. Market Size by 2033 | USD 3.93 Billion |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Segments Covered | By Device, By Application, and By End-use |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Cardiac Arrhythmia Monitoring Devices Market Dynamics
Drivers:
Several drivers propel the growth of the cardiac arrhythmia monitoring devices market. Technological advancements, such as the integration of artificial intelligence (AI) and machine learning algorithms into monitoring devices, enhance their diagnostic accuracy and efficiency. Moreover, the growing adoption of remote patient monitoring solutions enables real-time tracking of cardiac rhythms, facilitating timely intervention and reducing hospital admissions. Additionally, the increasing prevalence of risk factors such as hypertension, obesity, and diabetes further boosts the demand for monitoring devices, as early detection and management of arrhythmias become imperative in high-risk populations.
Opportunities:
The cardiac arrhythmia monitoring devices market presents numerous opportunities for growth and innovation. Expanding product portfolios to include wearable and implantable monitoring devices allows manufacturers to cater to diverse patient needs and preferences. Furthermore, partnerships and collaborations between healthcare providers, technology companies, and research institutions facilitate the development of advanced monitoring solutions and improve patient access to innovative technologies. Leveraging telemedicine and telehealth platforms for remote monitoring and consultation opens up new avenues for reaching underserved populations and improving healthcare delivery in remote or rural areas.
Challenges:
Despite the promising growth prospects, the cardiac arrhythmia monitoring devices market faces certain challenges. Regulatory hurdles and compliance requirements pose barriers to market entry for new entrants and can delay the approval and commercialization of innovative devices. Moreover, concerns regarding data security and patient privacy in remote monitoring systems need to be addressed to build trust among users and healthcare providers. Additionally, the high cost of advanced monitoring devices and limited reimbursement coverage in some regions may hinder market growth, especially in low- and middle-income countries where affordability remains a significant concern. Overcoming these challenges requires collaboration among stakeholders to develop cost-effective solutions and ensure equitable access to cardiac arrhythmia monitoring technologies.
Read Also: Artificial Organs Market Size to Rise USD 60.82 Billion by 2033
Recent Developments
- In October 2023, The LUX-Dx II+ Insertable Cardiac Monitor (ICM) System, developed by Boston Scientific, is a next-generation insertable monitor intended for long-term arrhythmia monitoring related to disorders like syncope, cryptogenic stroke, and atrial fibrillation (AF).
- In July 2022, Xplore Lifestyle introduced a smartwatch with medical-grade features for continuous cardiac health monitoring. This watch, created in collaboration with the Israeli company Cardiac Sense, is billed as the world’s first medical-grade continuous monitoring gadget in the shape of a watch. This medical gadget may distinguish between an arrhythmia, an abnormal or irregular heartbeat, and a normal heart rhythm, particularly atrial fibrillation (AFib).
Cardiac Arrhythmia Monitoring Devices Market Companies
- Applied Cardiac Systems
- AliveCor
- Biotronik
- Biotricity
- GE Healthcare
- iRhythm Technologies
- Koninklijke Philips N.V
- Medtronic plc
- Nihon Kohden Corporation
- St. Jude Medical (Abbott Laboratories)
- Spacelabs Healthcare (OSI Systems, Inc.)
- Welch Allyn (Hillrom Services, Inc.)
Segments Covered in the Report
By Device
- Holter Monitor
- Event Recorder
- Mobile Cardiac Telemetry
- Implantable Cardiac Monitor
- Electrocardiogram (ECG) Monitor
- Others
By Application
- Tachycardia
- Atrial Tachycardia
- Ventricular Tachycardia
- Bradycardia
- Premature Contraction
- Others
By End-use
- Hospitals and Clinics
- Diagnostic Centers
- Ambulatory Surgical Centers
- Homecare Settings
- Others
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
Blog: https://www.dailytechbulletin.com/
Blog: https://www.autoindustrybulletin.com/
- Liquid Handling Systems Market Size Expected to Hit USD 6.94 Billion by 2034 Driven by Rising Demand in Clinical Diagnostics and Genomics - September 26, 2025
- Quantum Dot Materials and Technologies Market to Surpass USD 46.4 Billion by 2034, Expanding at a CAGR of 22.64% - September 9, 2025
- Infrastructure For Business Analytics Market Size to Surpass USD 166.57 Bn by 2034, Growing at 9.05% CAGR - September 8, 2025
