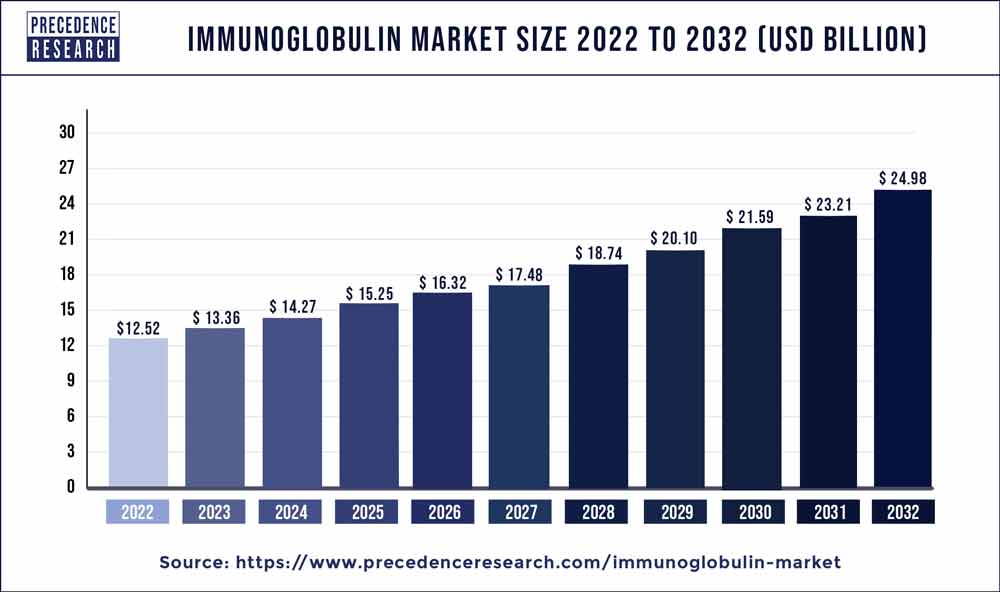
The immunoglobulin market size is projected to touch USD 24.98 billion by 2032 from USD 13.36 billion in 2023, growing at a CAGR of 7.20% during the forecast period 2023 to 2032.
Key Takeaways
- North America contributed more than 46% market share in 2022.
- Asia-Pacifica is estimated to witness the fastest CAGR between 2023 and 2032.
- By application, the hypogammaglobulinemia segment has held the largest market share of 38% in 2022.
- By application, the immunodeficiency disease segment is anticipated to grow at a remarkable CAGR of 9.1% between 2023 and 2032.
- By product, the IgA segment generated over 41% of market share in 2022.
- By product, the IgG segment is expected to expand at the fastest CAGR over the projected period.
- By mode of delivery, the Intravenous segment had the largest market share of 66% in 2022.
- By mode of delivery, the subcutaneous segment is expected to expand at the fastest CAGR over the projected period.
The Immunoglobulin market is a crucial segment within the pharmaceutical and biotechnology industries, primarily focusing on the production and distribution of immunoglobulin products. Immunoglobulins, also known as antibodies, play a pivotal role in the immune system, and the market encompasses a diverse range of therapeutic applications. These applications include treating immunodeficiency disorders, autoimmune diseases, and various infections. The market has experienced significant growth in recent years, driven by advancements in biotechnology, increasing prevalence of immunodeficiency disorders, and rising awareness about antibody therapies.
Region Snapshot
The Immunoglobulin market is globally distributed, with a strong presence in North America, Europe, Asia-Pacific, and other regions. North America, particularly the United States, holds a substantial market share, attributed to a well-established healthcare infrastructure, high healthcare expenditure, and a growing patient population. Europe follows closely, with countries like Germany and the United Kingdom contributing significantly to the market. The Asia-Pacific region is emerging as a key player, driven by increasing healthcare investments, a rising geriatric population, and a growing awareness of immunoglobulin therapies.
Get a Sample: https://www.precedenceresearch.com/sample/3646
Growth Factors:
Several factors contribute to the growth of the Immunoglobulin market, including a surge in research and development activities, technological advancements in protein engineering, and the expanding scope of immunotherapy. Additionally, the growing incidence of immunodeficiency disorders, autoimmune diseases, and infectious conditions fuels the demand for immunoglobulin products. The market benefits from the increasing adoption of intravenous immunoglobulin (IVIG) and subcutaneous immunoglobulin (SCIG) therapies, offering convenient treatment options for patients.
Immunoglobulin Market Scope
| Report Coverage | Details |
| Growth Rate from 2023 to 2032 | CAGR of 7.20% |
| Market Size in 2023 | USD 13.36 Billion |
| Market Size by 2032 | USD 24.98 Billion |
| Largest Market | North America |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Segments Covered | By Application, By Product, and By Mode of Delivery |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Drivers:
Key drivers propelling the Immunoglobulin market forward include the rising prevalence of primary immunodeficiency disorders (PIDD) and autoimmune diseases. The expanding applications of immunoglobulin in neurological disorders, hematology, and infectious diseases also contribute to market growth. Moreover, the approval of novel immunoglobulin products by regulatory authorities stimulates market dynamics. The increasing use of immunoglobulins in off-label indications further amplifies the market’s momentum.
Opportunities:
The Immunoglobulin market presents numerous opportunities for industry players, especially in exploring untapped markets in developing regions. Innovations in manufacturing processes, such as recombinant technology, open avenues for novel product development. Collaborations between pharmaceutical companies and research institutions enhance research capabilities, potentially leading to the discovery of new therapeutic applications. Furthermore, a growing emphasis on personalized medicine and targeted therapies creates opportunities for tailoring immunoglobulin treatments to specific patient profiles.
Challenges:
Despite its growth, the Immunoglobulin market faces challenges, including the high cost associated with immunoglobulin therapies, which may limit accessibility for some patient populations. Regulatory complexities and stringent approval processes pose obstacles for market entry. Additionally, potential side effects and adverse reactions associated with immunoglobulin administration require careful monitoring and management, adding a layer of complexity to treatment protocols.
Competition Landscape:
The Immunoglobulin market is characterized by intense competition among key players such as CSL Behring, Grifols, Kedrion Biopharma, Octapharma, and Takeda Pharmaceutical Company. These companies engage in strategic initiatives such as mergers, acquisitions, and collaborations to strengthen their market position. Continuous research and development efforts focus on improving product efficacy, safety profiles, and exploring new therapeutic indications. The competitive landscape is dynamic, with companies vying for a larger market share through innovation and geographical expansion.
Read Also: Genitourinary Drugs Market Size To Rake USD 32.27 Bn By 2032
Recent Developments
- In 2023, Everest Medicines gained approval from the Hainan Medical Products Administration for clinical use of Nefecon, a primary immunoglobulin A nephropathy (IgAN) drug, in China. This milestone marks progress in addressing IgAN, a condition affecting the kidneys, enhancing treatment options for patients in the region.
- In 2022, Grifols secured approval from European Union member state health authorities and the United Kingdom for XEMBIFY, a groundbreaking 20% subcutaneous immunoglobulin (SCIG). This innovative treatment is authorized for addressing primary and specific secondary immunodeficiency, marking a significant advancement in therapeutic options for these conditions.
- In 2021, Octapharma USA’s investigational new drug (IND) application for a phase three clinical trial assessing the efficacy and safety of Octagam 10% [Immune Globulin Intravenous (Human)] therapy in severe COVID-19 patients received authorization from the U.S. Food and Drug Administration (FDA).
Immunoglobulin Market Players
- Grifols, S.A.
- Shire
- CSL Behring
- Kedrion S.p.A.
- Octapharma
- Baxter International Inc.
- Biotest AG
- Bio Products Laboratory Ltd. (BPL)
- China Biologic Products Holdings, Inc.
- LFB SA
- Cangene Corporation
- Bharat Serums and Vaccines Limited
- Takeda Pharmaceutical Company Limited
- Bio Products Laboratory Ltd. (BPL)
- China National Pharmaceutical Group Corporation (Sinopharm)
Segments Covered in the Report
By Application
- Hypogammaglobulinemia
- Chronic Inflammatory Demyelinating Polyneuropathy
- Immunodeficiency Disease
- Myasthenia Gravis
- Multifocal Motor Neuropathy
- Idiopathic Thrombocytopenic Purpura
- Inflammatory Myopathies
- Specific Antibody Deficiency
- Guillainâ€Barré Syndrome
- Others
By Product
- IgA
- IgG
- IgM
- IgE
- IgD
By Mode of Delivery
- Intravenous
- Subcutaneous
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
Blog: https://www.dailytechbulletin.com/
Blog: https://www.autoindustrybulletin.com/
- Photodynamic Therapy Market Size to Rake USD 8.42 Bn by 2033 - February 5, 2024
- Image Recognition Market Size to Attain USD 166.01 Bn by 2033 - February 5, 2024
- Hydrogen Storage Tanks and Transportation Market Report 2033 - February 5, 2024
