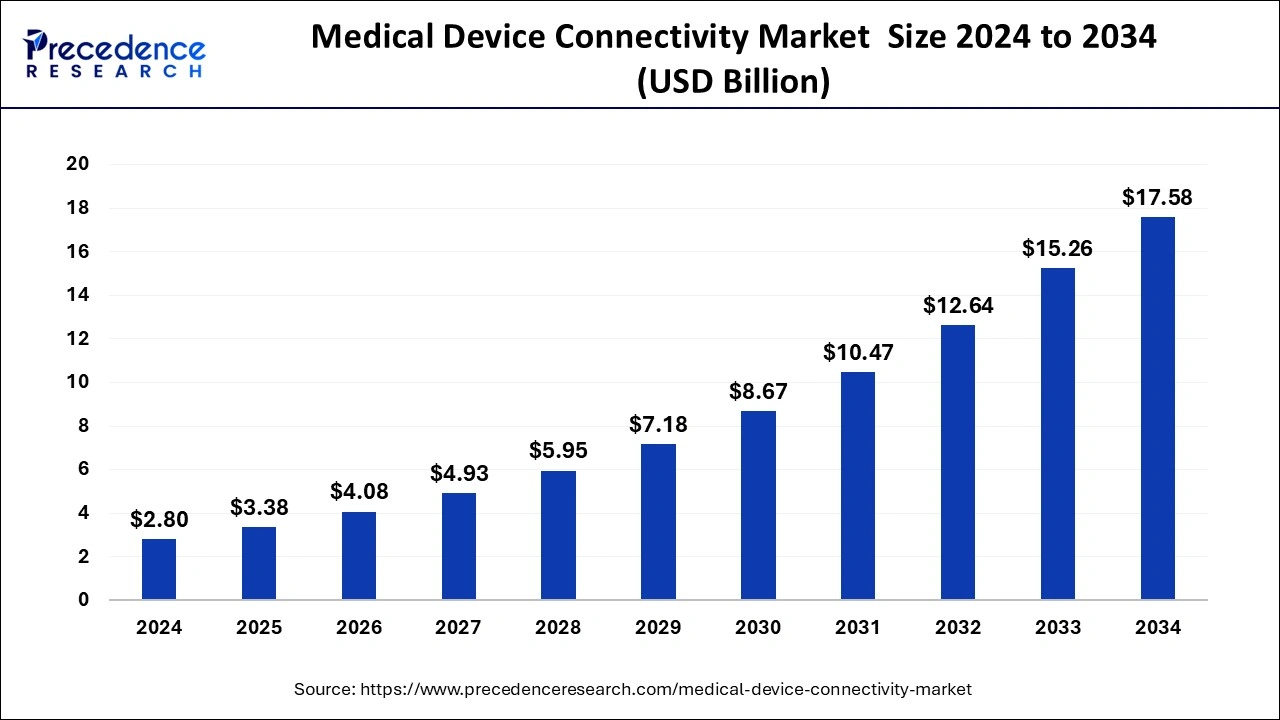
The global medical device connectivity market size was valued at and is anticipated to reach around USD 15.26 billion by 2033 from USD 2.32 billion in 2023, growing at a CAGR of 20.74% from 2024 to 2033.
Key Points
- North America has contributed 36% of market share in 2023.
- Asia Pacific is estimated to expand the fastest CAGR of 25.7% between 2024 and 2033.
- By components, in 2023, the wireless segment held the highest market share of 48%.
- By components, the wired segment is anticipated to witness rapid growth at a significant CAGR during the projected period.
- By application, the hospitals segment has held the biggest market share in 2023.
- By application, the ambulatory care centers segment is anticipated to witness significant growth over the projected period.
The medical device connectivity market has witnessed substantial growth in recent years, driven by the increasing adoption of digital healthcare solutions and the need for efficient data management in healthcare facilities. Medical device connectivity refers to the ability of medical devices to securely exchange data with electronic health records (EHR) systems, healthcare providers, and other medical devices. This connectivity enables real-time monitoring, remote patient management, and streamlined workflows, ultimately improving patient outcomes and operational efficiency in healthcare settings.
Get a Sample: https://www.precedenceresearch.com/sample/3996
Growth Factors:
Several factors contribute to the growth of the medical device connectivity market. One of the primary drivers is the rising demand for integrated healthcare solutions that enable seamless data exchange between various medical devices and healthcare IT systems. Additionally, the growing prevalence of chronic diseases, aging populations, and the need for remote patient monitoring have fueled the adoption of medical device connectivity solutions. Moreover, government initiatives promoting the use of electronic health records and interoperability standards further propel market growth.
Region Insights:
The medical device connectivity market exhibits significant regional variations in terms of adoption and market dynamics. North America dominates the market, driven by the presence of advanced healthcare infrastructure, high healthcare expenditure, and strong government support for healthcare IT initiatives. Europe follows closely, with countries like Germany and the UK leading in healthcare digitization efforts. Asia Pacific is emerging as a lucrative market, fueled by increasing investments in healthcare infrastructure, growing healthcare IT adoption, and rising awareness about the benefits of medical device connectivity solutions.
Medical Device Connectivity Market Scope
| Report Coverage | Details |
| Growth Rate from 2024 to 2033 | CAGR of 20.74% |
| Global Market Size in 2023 | USD 2.32 Billion |
| Global Market Size by 2033 | USD 15.26 Billion |
| U.S. Market Size in 2023 | USD 580 Million |
| U.S. Market Size by 2033 | USD 3,850 Million |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Segments Covered | By Components and By Application |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Medical Device Connectivity Market Dynamics
Drivers:
Several key drivers are driving the growth of the medical device connectivity market. One of the primary drivers is the need for improved patient care and clinical outcomes. Medical device connectivity enables healthcare providers to access real-time patient data, facilitating timely interventions and personalized care. Moreover, the implementation of telehealth and remote monitoring solutions has accelerated the adoption of medical device connectivity, especially in the wake of the COVID-19 pandemic. Additionally, the shift towards value-based care models and the focus on interoperability standards further drive market growth.
Opportunities:
The medical device connectivity market presents several opportunities for growth and innovation. One such opportunity lies in the integration of artificial intelligence (AI) and machine learning (ML) technologies with medical device connectivity solutions. AI-driven analytics can help healthcare providers derive actionable insights from vast amounts of patient data, leading to more informed decision-making and improved clinical outcomes. Furthermore, the increasing adoption of wearable devices and IoT-enabled medical devices opens up new avenues for expanding the scope of medical device connectivity, enabling continuous monitoring and preventive care.
Restraints:
Despite the promising growth prospects, the medical device connectivity market faces certain challenges and restraints. One of the key challenges is the issue of data security and privacy concerns. The interconnected nature of medical devices increases the risk of cybersecurity threats and unauthorized access to sensitive patient information. Ensuring robust data encryption, access controls, and compliance with regulatory standards is crucial to addressing these security challenges. Moreover, interoperability issues among different medical devices and healthcare IT systems pose challenges to seamless data exchange and integration, hindering the widespread adoption of medical device connectivity solutions. Regulatory complexities and reimbursement challenges further impede market growth, requiring stakeholders to navigate through stringent regulatory requirements and demonstrate the clinical and economic value of medical device connectivity solutions.
Read Also: Cord Blood Banking Market Size to Attain USD 27.55 Bn by 2033
Recent Developments
- In October 2023, Philips unveiled new interoperability features aimed at providing hospitals with a comprehensive patient health overview for enhanced monitoring and care coordination. By integrating Philips Capsule Medical Device Information Platform (MDIP) with Philips Patient Information Center iX (PIC iX), the company offers a unique patient monitoring ecosystem, bringing together diverse medical devices and systems onto a single interface. This interoperability allows clinicians to access streaming data from various device manufacturers on an open, secure platform, offering a new clinical perspective.
- In February 2023, the province of Nova Scotia collaborated with Nova Scotia Health Authority (NSHA) and IWK Health (IWK) to implement an integrated electronic care record system across the province, facilitated by Oracle Cerner. This technology aims to improve healthcare professionals’ access to patient information.
- In 2021, Koninklijke Philips N.V. entered into an agreement to acquire Capsule Technologies Inc., a leading provider of medical device integration and data technologies to healthcare organizations. This strategic move was intended to bolster Philips’ position in delivering connectivity solutions for patient care management within hospital settings.
- In 2021, Masimo Corporation introduced iSirona, a connectivity solution designed to integrate with electronic medical records (EMRs), surveillance monitoring, alarm management, mobile notifications, smart displays, and analytics.
Medical Device Connectivity Market Companies
- Cerner Corporation (U.S.)
- Medtronic (Ireland)
- Masimo (U.S.)
- Koninklijke Philips N.V. (Netherlands)
- General Electric (U.S.)
- Stryker (U.S.)
- iHealth Labs Inc., (U.S.)
- Cisco Systems, Inc. (U.S.)
- Lantronix Inc. (U.S.)
- Infosys Limited (India)
- Silicon & Software Systems Ltd. (Ireland)
- Hill-Rom Services Inc. (U.S.)
- Silex Technology America, Inc (Japan)
- Digi International Inc. (U.S.)
- Baxter (U.S.)
- TE Connectivity (Switzerland)
- Bridge-Tech (U.S)
- MediCollector (U.S.)
Segments Covered in the Report
By Components
- Wireless
- Wired
- Hybrid Technologies
By Application
- Hospitals
- Home Healthcare Centers
- Diagnostic Centers
- Ambulatory Care Centers
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
Blog: https://www.dailytechbulletin.com/
Blog: https://www.autoindustrybulletin.com/
- Liquid Handling Systems Market Size Expected to Hit USD 6.94 Billion by 2034 Driven by Rising Demand in Clinical Diagnostics and Genomics - September 26, 2025
- Quantum Dot Materials and Technologies Market to Surpass USD 46.4 Billion by 2034, Expanding at a CAGR of 22.64% - September 9, 2025
- Infrastructure For Business Analytics Market Size to Surpass USD 166.57 Bn by 2034, Growing at 9.05% CAGR - September 8, 2025
